Curriculum

Biotechnology
Biotechnology has brought about the discovery and development of a new generation of human therapeutics. Advancements in both cellular and molecular biology have allowed scientists to identify and develop a host of new medicines for patients with serious illness. Biotechnology provides the tools and techniques for modern pharmaceutical research and drug development, and it is critical that future citizens are knowledgeable about this field.
The Program and Curriculum
The ABE program integrates a curriculum that allows students to explore the steps involved in creating biotechnology therapies. Aligned with the core biology curriculum, the program supports the larger goal of fostering scientific literacy. In addition to the curriculum and teacher professional development to understand the lab protocols and science, participating teachers receive a loaned kit, free of charge, with research-grade equipment and supplies that allow students to participate in advanced science laboratories.
The Labs and Materials
The ABE labs parallel some of the important steps taken by the biotechnology industry to develop medicines to treat a variety of diseases. The labs incorporate core technologies used by scientists in the discovery of human therapeutics, so that students will better understand the role of biotechnology and the potential impact of this industry on our future. In addition, by engaging in this program, students will be more motivated to understand the underlying science concepts and perhaps even pursue careers in science.
CHAPTER 1
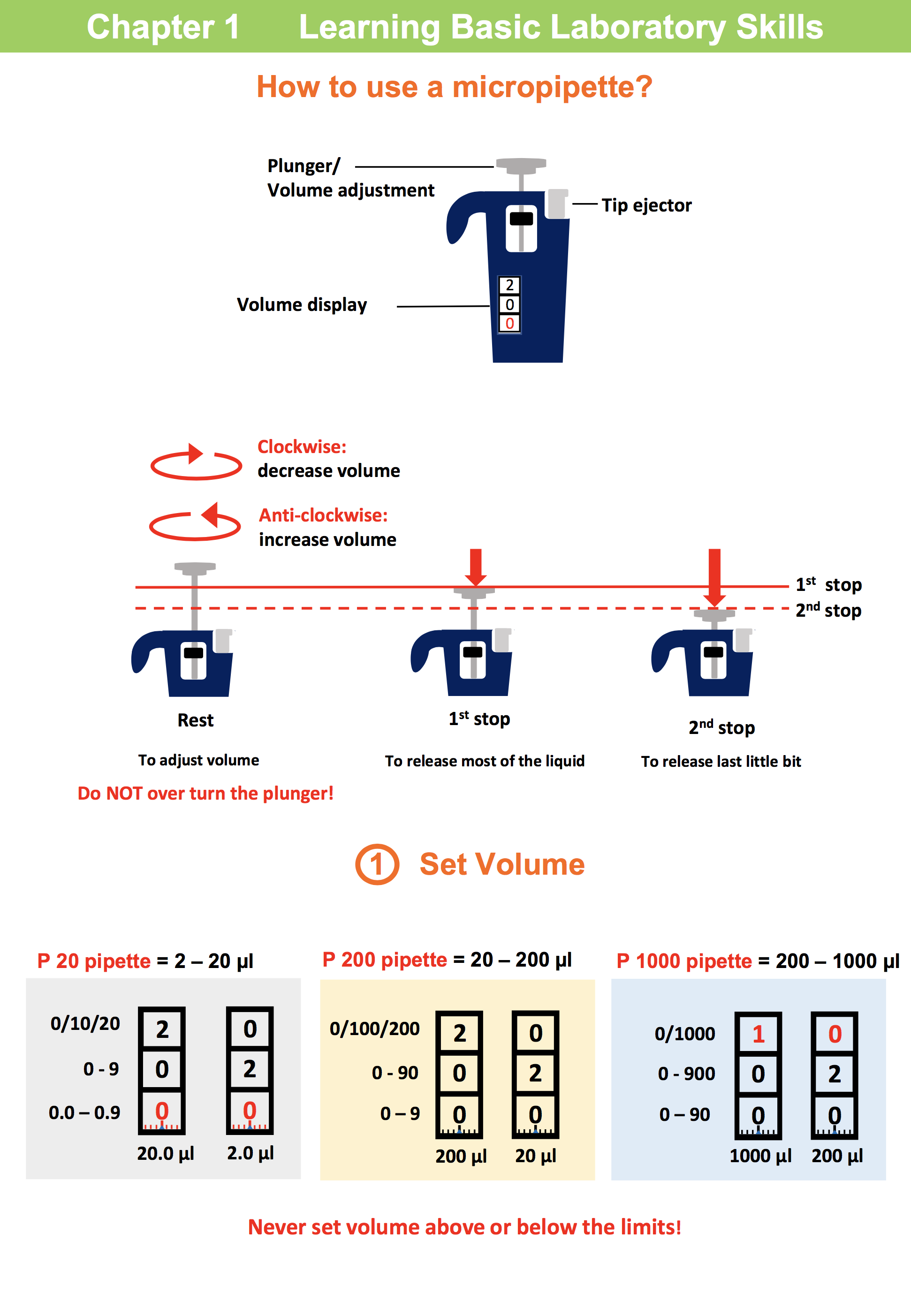
In chapter 1 , students learn the basic techniques essential to biotechnology, such as micro-pipettes, gel electrophoresis systems and centrifuges.
Lab 1.1 Basic pipetting and serial dilution
Lab 1.2 Dye separation by gel electrophoresis

CHAPTER 2
The most widely used method in biotechnology is the polymerase chain reaction (PCR). PCR provides a way to quickly and accurately identify a specific DNA sequence and then make multiple copies of that sequence. In this chapter, students learn about the multiple uses of PCR and then carry out the PCR to confirm the identity of the plasmid provided.
In Chapter 2, students use PCR to amplify sections of the non-recombinant plasmid and the recombinant plasmid with red fluorescent protein gene (rfp). The sizes of the amplified sequences in each plasmid vary and therefore can be used to identify the two plasmids.
Lab 2.1 Checking plasmid with PCR
Lab 2.2 Confirmation by Gel Electrophoresis (PCR products)

CHAPTER 3
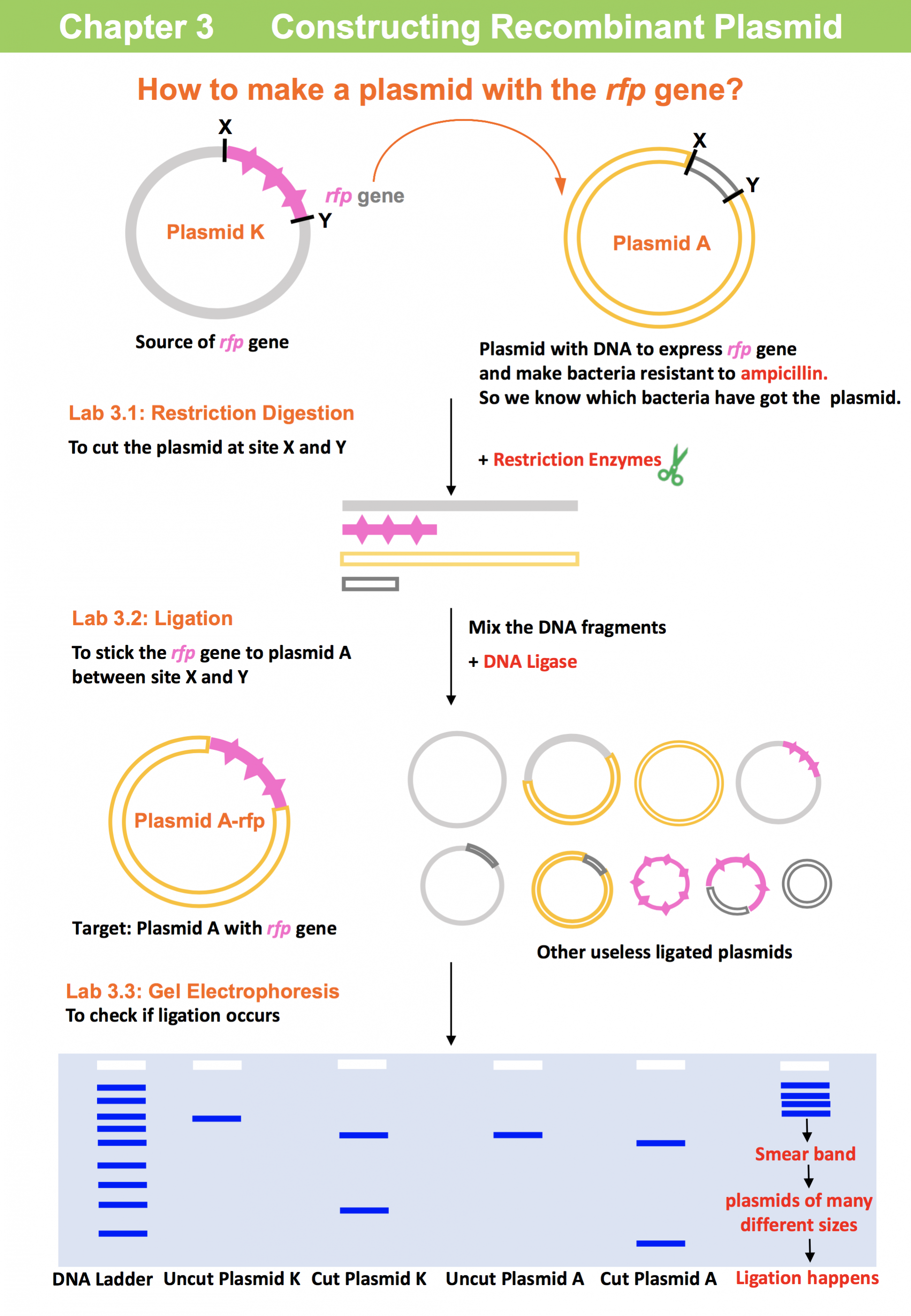
The purpose of chapter 3 is to produce the recombinant plasmid A with the red fluorescent protein gene (rfp), which can make the red fluorescent protein in bacteria. Students use restriction enzymes to cut two plasmids, which will generate DNA fragments, i.e. restriction digestion. The fragments are then linked by performing the ligation reaction. The lengths of the products from the restriction digest and the ligation are examined by gel electrophoresis.
Lab 3.1: Cutting the two plasmids (Restriction Digestion)
Lab 3.2: Putting the rfp gene into the plasmid (Ligation)
Lab 3.3: Confirmation by Gel Electrophoresis (Digestion and Ligation products)
CHAPTER 4
In chapter 4, students will carry out another step of the gene cloning process, which is to transform E. coli bacteria with the recombinant plasmid.
Lab 4: Transforming bacteria with recombinant plasmid
